But regardless of how many PK A values there are, there could be five. 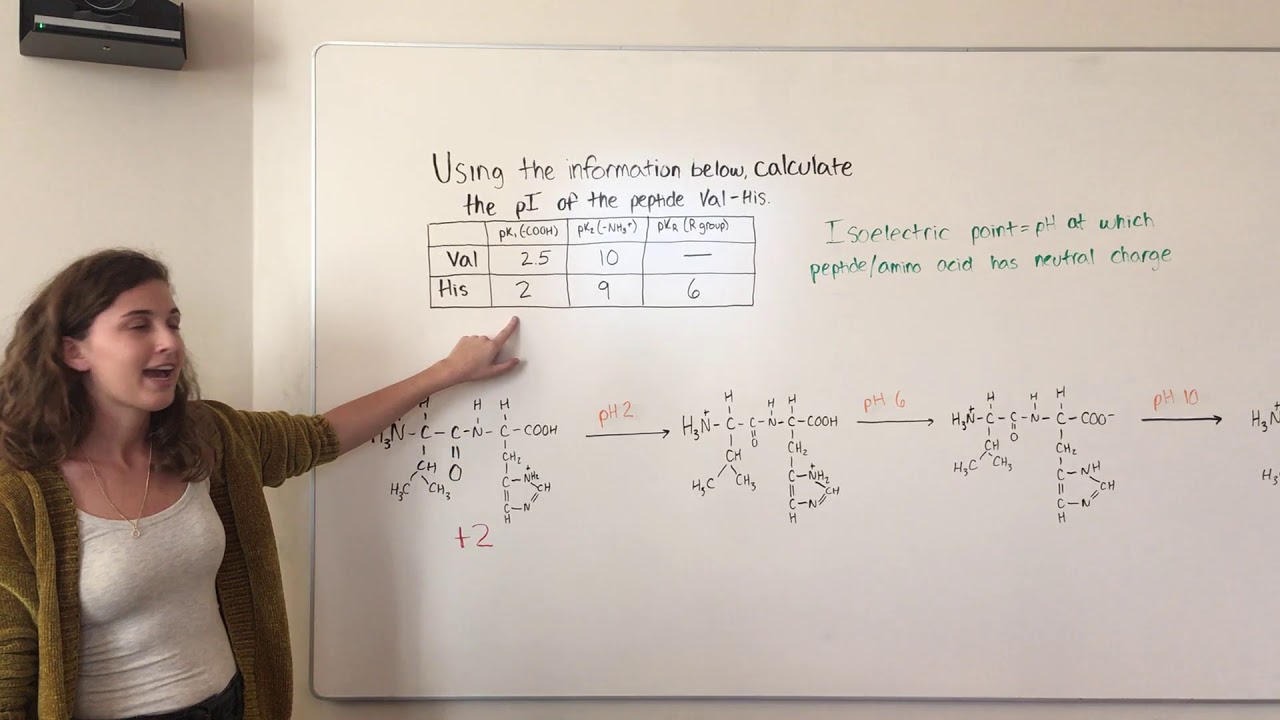
And so here we're showing Onley four p k a values. Now step number two is going to be to order the PK a values from the smallest to the greatest. And so if you know this pneumonic and how it works, then you'll know those seven amino acid with ionized able are groups and you'll be set to go with step number one. And again, the first step is just gonna be to know to know how the amino acids are groups ionized and really all this is saying is that we need to know the seven amino acids with ionized herbal are groups and recall that are pneumonic to help us remember those seven amino acids is just yucky crazy dragons eat nights riding horses. So let's take a look at the four steps that we need to follow to calculate the ice electric point of a peptide. And so this little chart here, this little diagram might look familiar to you because it's really the same one from our previous lesson. And so the second idea that I really wanna emphasize is just this idea that the ISO electric point of a peptide follows similar steps for calculating the ISO electric point of amino acids with Ayan Izabal are groups. And so when we're doing our practice problems, we're gonna be using a specific set of PKs for amino acid residues. But just in case you should double check to make sure that your professor doesn't want you to use a specific set of P. K s for amino acid residues and free amino acids, then you're good to go, and you don't have to worry about this. And so, if you're professor uses the same set of P. And because we know that the micro environment is capable of shifting the PK a values of residues, you need to make sure that you're using the correct set of PK A values specifically the set of PK a values for amino acid residues and not for PK is of free amino acids. And so the troop EI values can Onley be experimentally determined. And this is because of the unique micro environment that each residue has in a peptide. And this is the idea that the is electric point and the net charge of a peptide can both on Lee be estimated. And the first is a similar idea to the one that we talked about when we were calculating the net charge of a peptide. And so, before we get to our example below, there's really two ideas that I really wanna emphasize. And the same is gonna apply for calculating the is electric point of a peptide. And so all this is saying is that whenever we're calculating the ISO electric point, we're only gonna average two p k s regardless of how many p K s were given. S for the two ionization involving the neutral species. So recall from our previous lesson videos on the is electric point that the is electric point or the P I calculation is always just gonna be the midpoint or the average between the two p. In this video, we're gonna talk about how to calculate the YSL electric point of a peptide. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
